 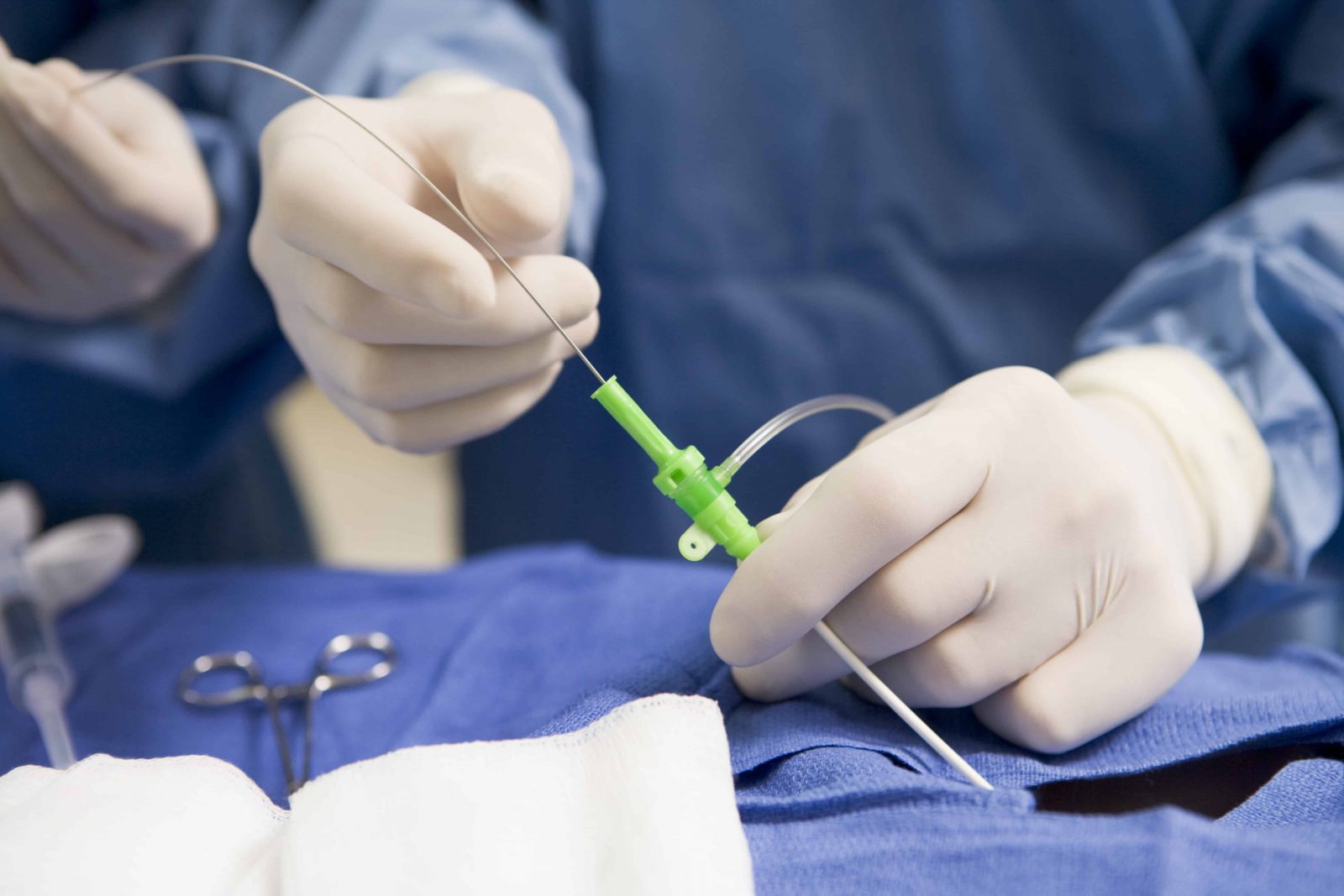
Price Action: TFX shares are up 0.29% at $237.98 on the last check Monday. better level of fitness, including strength, stamina (able to exercise or do an activity longer), and being able to do your daily activities better quality of. There have been 18 reported injuries and no deaths. Single-use peripheral intravascular device designed to permit access to the peripheral vascular system. to be closed using cardiac catheterization or minimally invasive surgical techniques. Teleflex/Arrow International reports 83 complaints related to this issue. Arrow Endurance Extended Dwell Peripheral Catheter System. arrhythmias heart muscle weakness decreased endurance (exercise. The issue may cause serious injury, including blockage of blood vessels, inadequate blood flow, injury to blood vessel walls, blood clots, blockage of the lung arteries (pulmonary embolism), heart attack, or death. The FDA writes that if the catheter separates while in a blood vessel, the catheter fragments could be left in the bloodstream and may migrate to other places in the body.Īlso Read: Recall Alert: Teleflex Recalls Rüsch Endotracheal Tubes Due to Connector Issues. The ARROW Endurance Extended Dwell Peripheral Catheter System allows healthcare providers access to a patient’s peripheral vascular system for short-term use to sample blood, monitor blood pressure, or administer fluids, blood, and blood products. The FDA has identified this as a Class I recall, the most serious type of recall. Conclusions: Short-term intravenous vancomycin can be safely and cost-efficiently administered in the deep vessels of the upper arm using the midline study device. Insertion costs were 90.00 less per insertion in the midline group. All rights reserved.Teleflex Incorporated (NYSE: TFX) and its subsidiary Arrow International are recalling the ARROW Endurance Extended Dwell Peripheral Catheter System after reports of catheter separation or leakage. One suspected catheter-associated bloodstream infection did occur in the PICC group. Midline midline catheter venous access devices.Ĭopyright © 2016 Elsevier Inc. Its utilization in the ED in patients deemed to require prolonged hospitalization or to have difficult-to-access peripheral vasculature could reduce cost and risk to patients. The MC is a versatile venous access device with a low complication rate, long dwell time, and high rate of first-attempt placement. Placement of a MC includes modified Seldinger and accelerated, or all-in-one, Seldinger techniques with or without ultrasound guidance, with a high rate of first-attempt success. Cost of insertion of a MC has been cited as comparable to three PIVs, and their use has been associated with significant cost savings when placed to avoid prolonged central venous access with CVCs or in patients with difficult-to-access peripheral veins. The average dwell time of a MC is reported as 7.69-16.4 days, which far exceeds PIVs (2.9-4.1 days) and is comparable to PICCs (7.3-16.6 days). Catheter, peritoneal, long-term indwelling - Product Code FJS: Product: Flex-Neck Catheter External Repair Kit REF CE-1400 LOT CE-1400/A: Code Information: Catalog Number: CE-1400/A UDI-DI Code: 00884450170069 Lot Number: H2586496: Recalling Firm/ Manufacturer: Merit Medical Systems, Inc. Teleflex, and their subsidiary Arrow International, are recalling the ARROW Endurance Extended Dwell Peripheral Catheter System after reports of catheter. Midline catheters (MC) offer a comparable rate of device-related bloodstream infection to standard peripheral intravenous catheters (PIV), but with a significantly lower rate than peripherally inserted central catheters (PICC) and central venous catheters (CVC) (PIV 0.2/1000, MC 0.5/1000, PICC 2.1-2.3/1000, CVC 2.4-2.7/1000 catheter days). To compare venous access device indications and complications, highlighting the use of midline catheters as a potentially cost-effective and safe approach for venous access in the ED. PMID: 32618955 DOI: 10.1097/NAN.0000000000000376 Abstract Maintaining and restoring patency in midline catheters has been a significant issue in the hospitalized patient requiring a multitude of infusates and frequent blood specimen collection. Appropriate device selection is warranted on initial patient contact to minimize risk and cost. The availability of trained personnel and dedicated teams using ultrasound-guided insertion techniques in technically difficult situations may also impact the selection. The patient's clinical status, ongoing need for laboratory investigation, and intravenous therapeutics guide the size, type, and placement of the catheter. Venous access in the emergency department (ED) is an often under-appreciated procedural skill given the frequency of its use. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
